If you find these posts interesting and think others might do so, spread the word.
In the last newsletter I gave a predicted outcome of the EMA review.
The decisions are likely to due to focus on CVST concerns, although the more general concern about thrombosis will need to be addressed.
The decisions are not a binary choice between stopping the use of AZ vaccine and continue use of AZ vaccine. Other options exist, such as noting the possible link of a rare adverse effect with the vaccine will exist, even without saying that the link is proven, while at the same time noting that the overall balance of benefit and harms still continues to be favourable.
There are options around further pharmacopidemiology studies that can be carried out to look at the risk of CVST, and continued analysis of clinical cases reported to look for aggravating risk factors and/or alternate explanations.
There will be decisions made about how to communicate potential risks to the public. This is always the tricky bit.
This turned out to be pretty accurate.
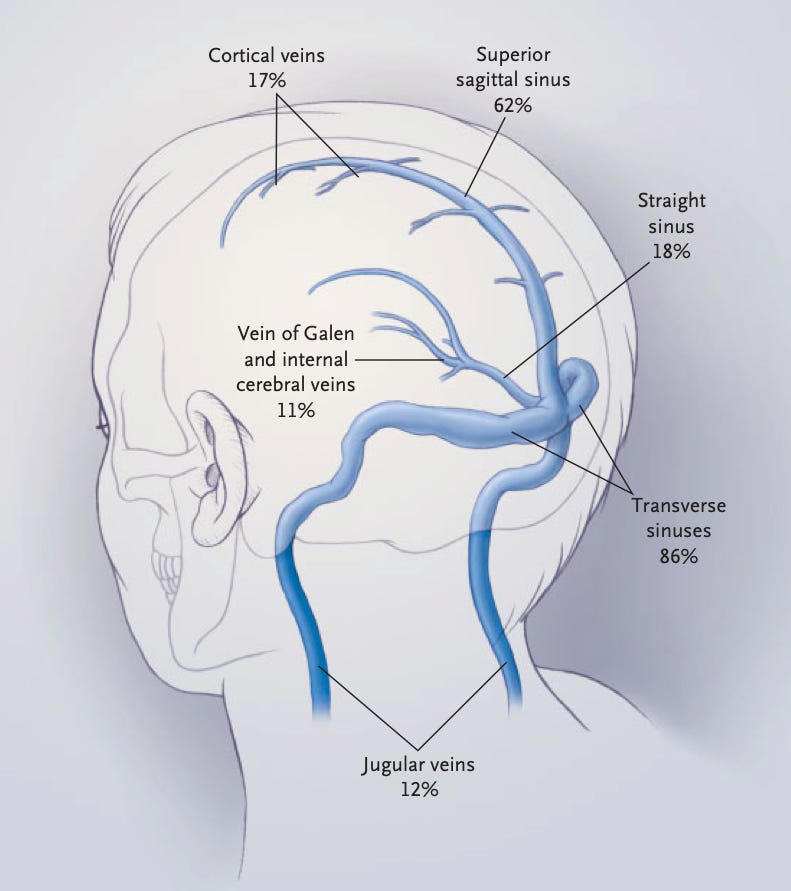
On Thursday both the MHRA and EMA came out with their review outcomes. The main focus was the issue that was highlighted by Norway and Germany around cerebral sinus venous thrombosis (CVST)1.
This can occur naturally, with an incidence of around 3-4 cases per million (A John Hopkins University source noted 5 cases per million). The Paul Erlich Institute reported 7 cases in 1.6 million vaccinations, which is just over 4 cases per million. Peter Arlett, Head of Pharmacovigilance and Epidemiology Department at the European Medicines Agency, at the press conference earlier in the week noted that epidemiology on such a rare condition isn’t that reliable, and that careful examination of all the clinical cases would be necessary.
The MHRA
The MHRA said that the available evidence didn't suggest a link with venous thromboembolism. They also carried out a detailed review of the 5 cases of CSVT reported in the UK (in 11 million vaccinated individuals, so less than 1 case per million). They noted that these clots could occur naturally, and concluded the link to the vaccine was unproven.
The balance of benefits of the AZ vaccine in preventing COVID-19 were confirmed, although they did suggest that:
Anyone with a headache that lasts more than 4 days or who have bruising beyond the site of vaccination after a few days seek medical attention.
The EMA
The EMA said (and you can watch the entire press conference if you wish) much the same. They noted the vaccine was not associated with any overall risk of blood clots, but did note that the vaccine might be linked to rare blood clots associated with thrombocytopenia (including CSVT). They noted that in 20 million people vaccinated in the UK and EEA (which also includes those states not in the EU, but covered by EMA) there had only been 18 cases of CVST. They also noted that there had been 7 cases of disseminated intravascular coagulation (DIC).
The Committee’s experts looked in extreme detail at records of DIC and CVST reported from Member States, 9 of which resulted in death. Most of these occurred in people under 55 and the majority were women. Because these events are rare, and COVID-19 itself often causes blood clotting disorders in patients, it is difficult to estimate a background rate for these events in people who have not had the vaccine. However, based on pre-COVID figures it was calculated that less than 1 reported case of DIC might have been expected by 16 March among people under 50 within 14 days of receiving the vaccine, whereas 5 cases had been reported. Similarly, on average 1.35 cases of CVST might have been expected among this age group whereas by the same cut-off date there had been 12. A similar imbalance was not visible in the older population given the vaccine.
They noted that ‘A causal link with the vaccine is not proven, but is possible and deserves further analysis.’
They also advised that
Recipients should be warned to seek immediate medical attention for symptoms of thromboembolism, and especially signs of thrombocytopenia and cerebral blood clots such as easy bruising or bleeding, and persistent or severe headache, particularly beyond 3 days after vaccination.
The Summary of Product Characteristics has now been updated.
As a side note, Peter Arlett suggested the disproportionate cases of CSVT in the EU compared to the UK might be because younger frontline workers (disproportionately women) were more likely to have been given the Pfizer vaccine in the UK since it was largely restricted to hospital sites due to the more complex storage issues.
What now?
The immediate drama over the AZ vaccine’s safety is over, with both EMA and the MHRA in close alignment on their decision making and science. The pause in European countries appears over, with a slight wrinkle in France which I’ll cover later.
Broadly the EMA and the MHRA have managed to stick with their scientific plans for managing the COVID-19 vaccine safety, and an important safety signal has been investigated. In addition, the EMA and the MHRA clearly worked closely together as is necessary. Even though a link is not proven, precautionary advice has been issued to warning both patients, and information healthcare staff (which I have already been using in my role as a vaccinator). Additional pharmacoepidemiological studies will be carried out to look at this safety signal. So the system of pharmacovigilance has broadly worked as it has meant to have worked.
EU state level decision making
The earlier pauses before the EMA review was completed still look to me like a poor set of decisions, and those EU countries that continued vaccinating in the face of rising COVID-19 cases look like they made the right decision. Given the rapidity of the EMA review, carrying on vaccinating would have been the safer option given the rarity of the event of concern.
There’s no point in having a carefully thought through safety plan for COVID-19 vaccines at an EU level that is meant to serve the dual purpose of protecting vaccine confidence and investigating real harm, if individual countries do their own ad hoc plans.
And amazingly, despite the EMA review, France is restricting the AZ vaccine to the over 55s, since most of the cases of CSVT and DIC were in those under 55 years of age.
To my mind the vaccine pauses and the French decision on under 55 year olds undermine EMA, and I am starting to wonder about the nature of the committees making these decisions. Are they always the group with the highest competency in pharmacovigilance? I’m not alone.


Montastruc isn’t the only clinical pharmacologist and world leading expert on pharmacovigilance in France, who has expressed such views.
The French decision was taken by HAS, which isn’t the body that normally oversees medicines in France (and whose stated functions don’t seem to fit pharmacovigilance). The French National Agency of Medicines and Health Product Safety, ANSM, was set up after some high profile safety scandals. It’s priorities were means to be safety and vigilance, so I am not sure why they weren't the ones advising the French government.
The German health Ministry halted the use of the AZ vaccine after advice from the Paul Erlich Institut, whose regulatory responsibility is around biological medicinal products (such as vaccines) in Germany, but not the Federal Institute for Drugs and Medical Devices (BfArM). I can’t help wondering if they might have come to a different decision around the pause, if they had consulted each other.
In contrast, Belgium seemed to have relied on its pharmacovigilance body FAMHP, which resisted the pressure to pause the AZ vaccine. They stuck with the calmer approach of EMA throughout the week, even to the extent of cheekily asking for an extra AZ vaccine from countries pausing the AZ vaccine to be sent to them.
Certainly, it appears that some individual states were struggling with making proportionate decisions in the face of concerns over the past week, and perhaps ought to consider reviewing their regulatory alignment on pharmacovigilance with EMA’s scientific processes and expertise in pharmacovigilance.
While the EU may have messed up vaccine procurement, they continue to have a world class medicines safety regulator that EU states should listen to.
Further reading
David Spiegalhalter and Anthony Masters Don’t Fear the AstraZenaca jab, the risks are minimal.
That’s all and stay safe.
Anthony
Don’t forget to report your suspected adverse effects from medicines and vaccines. In the UK, this means using the Yellow Card Scheme.